Cooper, A. I. and Bojdys, M. J. (2016). Two-dimensional carbon nitride material and method of preparation. WO2016027042 (A1).
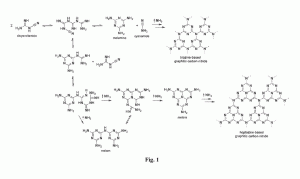
Graphitic carbon nitride has been prepared and its structure confirmed by extensive characterization. This material has useful electronic, in particular semiconducting, properties. Crystalline thin films have been prepared. Synthesis may be carried out by condensation of unsaturated carbon- and nitrogen- containing compound(s) in inert solvent such as a salt melt, forming graphitic carbon nitride at a gas-liquid or solid-liquid interface.
External Links: Patentscope, Espacenet, Google Patents


 Materials chemist
Materials chemist