Algara-Siller, G.; Severin, N.; Chong, S. Y.; Björkman, T.; Palgrave, R. G.; Laybourn, A.; Antonietti, M.; Khimyak, Y. Z.; Krasheninnikov, A. V.; Rabe, J. P.; Kaiser, U.; Cooper,* A. I.; Thomas, A.; Bojdys,* M. J. Angewandte Chemie International Edition 2014, 53, 7450–7455.
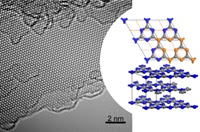
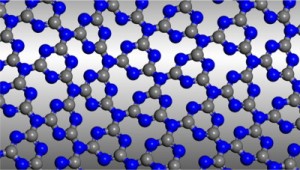
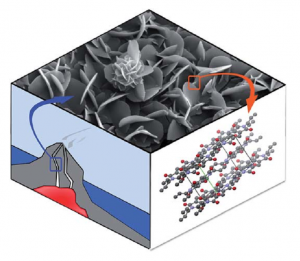
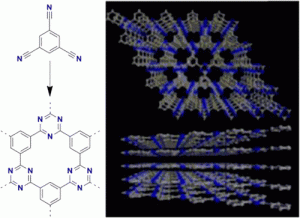
Graphitic carbon nitride has been predicted to be structurally analogous to carbon-only graphite, yet with an inherent bandgap. We have grown, for the first time, macroscopically large crystalline thin films of triazine-based, graphitic carbon nitride (TGCN) using an ionothermal, interfacial reaction starting with the abundant monomer dicyandiamide. The films consist of stacked, two-dimensional (2D) crystals between a few and several hundreds of atomic layers in thickness. Scanning force and transmission electron microscopy show long-range, in-plane order, while optical spectroscopy, X-ray photoelectron spectroscopy, and density functional theory calculations corroborate a direct bandgap between 1.6 and 2.0 eV. Thus TGCN is of interest for electronic devices, such as field-effect transistors and light-emitting diodes.
DOI: 10.1002/anie.201402191
This is the pre-peer reviewed version of the following article: Algara-Siller, G.; Severin, N.; Chong, S. Y.; Björkman, T.; Palgrave, R. G.; Laybourn, A.; Antonietti, M.; Khimyak, Y. Z.; Krasheninnikov, A. V.; Rabe, J. P.; Kaiser, U.; Cooper,* A. I.; Thomas, A.; Bojdys,* M. J. Angewandte Chemie International Edition 2014, 53, 7450–7455, which has been published in final form at [DOI: 10.1002/anie.201402191].