Veith,* G. M.; Baggetto, L.; Adamczyk, L. A.; Guo, B.; Brown, S. S.; Sun, X.-G.; Albert, A. A.; Humble, J. R.; Barnes, C. E.; Bojdys, M. J.; Dai, S.; Dudney, N. J. Chemistry of Materials 2013, 25, 503-508.
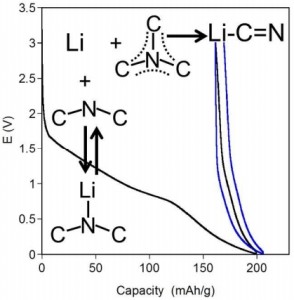
Lithiated graphitic carbon nitride (C3N4) was fabricated by electrochemical and solid-state reactions. The addition of Li to C3N4 results in a reaction between the Li and the graphite like C3N species in C3N4. This irreversible reaction leads to the formation of Li-CH=NR and Li-N=CR2 species which are detrimental to anode properties. Suitable nitrogen doped carbon structures for anode applications are shown to need high concentrations of pyridinic C-N-C terminal bonds and low concentrations of quaternary C3N species to boost electronic conductivity and reversibly cycle Li-ions.
DOI: 10.1021/cm303870x [Download]
Reprinted with permission from Veith, G. M.; Baggetto, L.; Adamczyk, L. A.; Guo, B.; Brown, S. S.; Sun, X.-G.; Albert, A. A.; Humble, J. R.; Barnes, C. E.; Bojdys, M. J.; Dai, S.; Dudney, N. J. Chemistry of Materials 2013, 25, 503-508. Copyright 2013 American Chemical Society.